Intermolecular Forces
|
|
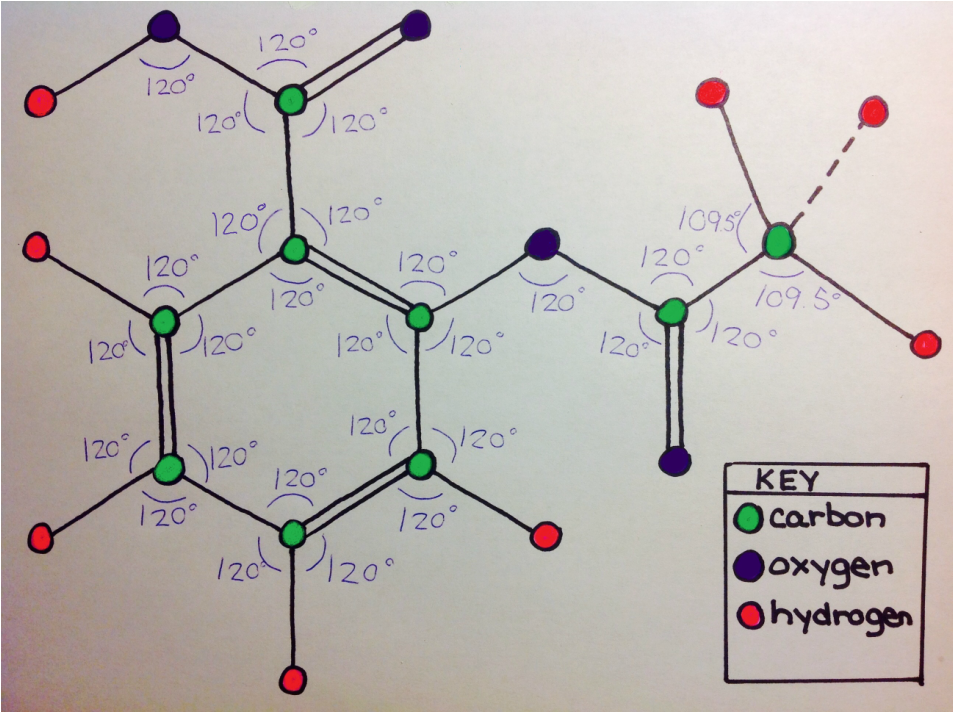
As seen to the left, the aspirin Molecule is adjacent to the water molecule in each picture. These images show that several intermolecular forces are at work between the two molecules.
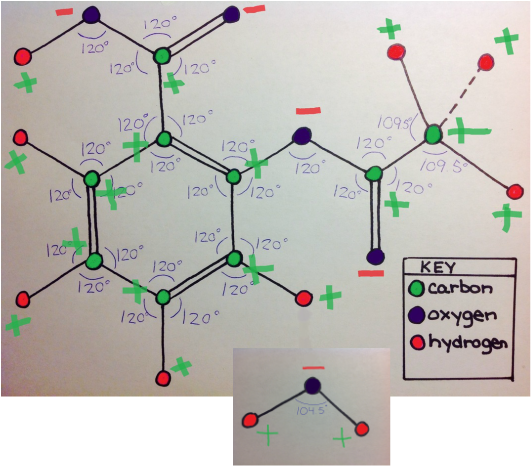
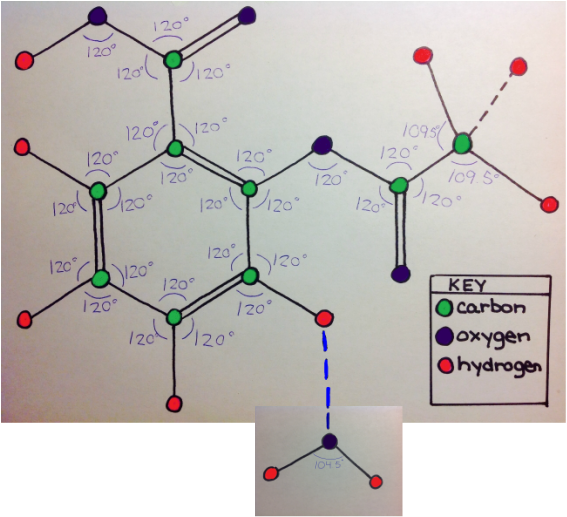
The first force is dispersion, which is a force that occurs between any two adjacent molecules. This can be seen in the top image. Because the aspirin molecule is adjacent to the water molecule, dispersion occurs. Dispersion would also occur with any other molecule adjacent to the aspirin molecule. The second force is dipole-dipole, which is an intermolecular force between any two polar molecules. This can be seen in the center image because each molecule is labeled with their charged to show that both molecules are polar. Because both water and acetylsalicylic acid are polar, dipole-dipole force is able to be present. This force will occur whenever the aspirin molecule is near another polar molecule. The last force is hydrogen bonding, which can occur between a hydrogen atom of a molecule and the oxygen, nitrogen, or fluorine. A possible example of hydrogen bonding can be seen by the blue dotted-line that bonds aspirin's hydrogen and water's oxygen in the bottom immage. Hydrogen bonding could occur between any of aspirin's hydrogen atoms with water's oxygen atom, and it could occur between a hydrogen atom of water with an oxygen atom of aspirin. Hydrogen bonding in aspirin could occur in any molecule with oxygen, hydrogen, nitrogen, or fluorine atoms. |